Tried and Tested Dr. Frei Protect Medical Masks
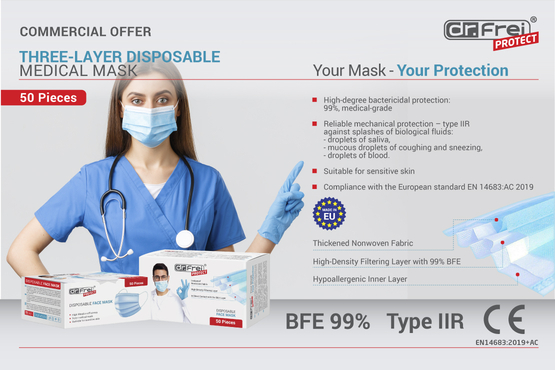
A commercial hit of this year, our three-layer disposable face masks, made in the EU, have lately been awarded a high category for protection in their class: medical masks of type IIR! Now officially recognized as medical devices, Dr. Frei Protect single-use facepieces are widening the scope of their application – adding to domestic, commercial, light-industrial, and industrial environments a medical setting as well.
Such a momentous accomplishment became possible after the mask specimens successfully passed strict testing procedures held in observance of European Standard EN 14683:2019+AC:2019. According to test directions, EUROLAB Laboratory carried out the textile analysis of Dr. Frei Protect disposable face mask in four categories: bacterial filtration efficiency, breathability (differential pressure), splash resistance and microbial cleanliness (bioburden). The resistance to penetration of splashes of liquid – which in real life are biological fluids like droplets of saliva, mucous sprays from coughing and sneezing or driblets of blood – turn out high enough to define our mask as type IIR. Accordingly, it may be used by healthcare professionals in an operating room or in other medical settings with similar requirements.
Moreover, the mask's microbial barrier also showed excellent high results at the level of 99% BFE (bacterial filtration efficiency). Finally, the material itself was put to the trials and scored zero on test for irritation and passed a skin sensitisation test with close to no visible change.
We invite our new and long-term partners to place bulk requests with our respective country managers and sales directors and wish all of us to stay well and protected!
Date: 2020-10-15We promise to send you only good news